CAR-T Platform
Immunotherapy has emerged as one of the most promising approaches for the treatment of cancer. Unlike traditional cancer treatments, immunotherapy harnesses the natural power of your immune system to fight the cancer. T cells, or T lymphocytes, are the armed forces of our immune system that are constantly circulating our bodies looking for foreign antigens. Upon recognition of the infected or cancerous cell, the T-cell becomes activated and initiates the immune response. Genetically modifying T cells with Chimeric Antigen Receptors (CARs) is the most pursued approach to generate tumor-specific T cells. CAR-T cells targeting tumor-associated antigens can be infused into patients to attack the tumor.
ProMab Biotechnologies Custom Chimeric Antigen Receptor Cell Engineering: A complete service, from antigen to clinically relevant solution.
ProMab Biotechnologies has developed many CAR-T products based on nearly 20 years' experience in hybridomas, monoclonal antibodies (rabbit, mouse, and human), recombinant proteins, and cancer stem cells. In addition, we hold over 8 years of experience in CAR-T research with an abundance of publications on CAR-T efficacy against cancer.
We offer an all-in-one service which includes services from antibody generation to CAR-T testing in vitro or in vivo with the potential for GMP CAR-T production. In addition, ProMab’s CAR-T platform is performed directly in the US and does not require outsourcing. Our new custom CAR-T service is designed to take advantage of the astonishing clinical breakthroughs of CAR-T cells against various hematological malignancies.
Our scientists can construct CARs based on your given antibody sequence or we can generate a mouse or fully human single-chain variable fragment (scFv). We have generated more than 30 types of CAR-T and CAR-NK cells targeting different tumor cells and cell lines expressing tumor-specific antigens that can be used with immunological modulators, tumor microenvironment invaders, or checkpoint inhibitors.
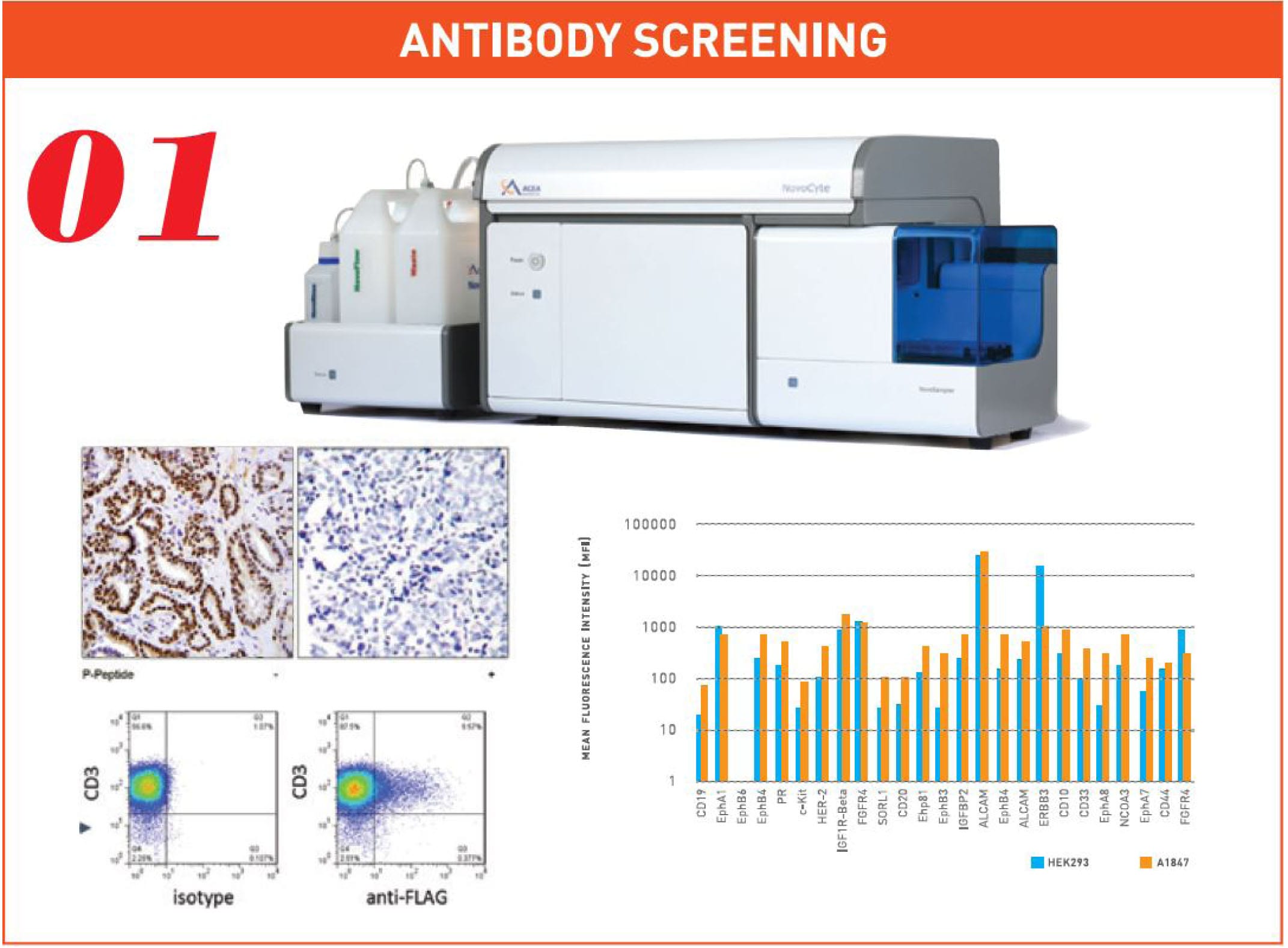
There are 8 necessary components to ProMab’s successful CAR-T platform. Firstly, our scientists generate the scFvs that recognize the unique cancer cell marker specifically and with optimal affinity. This is the key component of CAR as the antigen-binding capabilities are defined by the scFv, not the antigen. scFVs are engineered antibodies that are generated via the fusion of heavy and light-chain immunoglobulins to a short peptide linker. We offer two different methods of generating scFVs using the amino-terminal variable domain of the heavy and light chains (VH-VL).
The first method utilizes mouse monoclonal antibody development to generate the desired svFv. Firstly, our scientists synthesize and conjugate the desired antigen for several immunizations in mice. Next, the immunized mice plasma cells are fused with compatible myeloma cell lines to produce hybridoma cells which will be screened and bio-assayed for confirmation. The mRNA is then extracted from hybridoma cells for amplification and sequencing, and the resulting VH-VL will be reformatted with a linker to produce the desired scFv sequence.
The second method utilizes our human fab library to generate the desired scFv. The target antigen is screened against our extensive human Fab library to get the Fab binder sequence to the target. Once the VH-VL domain sequence is confirmed, our scientists insert a linker sequence between the specific immunoglobulin domains to generate the scFv sequence.
The successful scFv sequence will then be inserted into a lentiviral vector along with the CAR domain for molecular cloning and subsequent transduction in activated T-cells. The newly generated CAR-T cells will be subject to biological assays to confirm binding affinity and cytotoxicity against target cells. In addition, ProMab’s in vivo NSG/ NOG xenograft models are used to test the CAR-T cell’s anti-cancer activity in hematological and solid cancers.
Here we outline 8 components to a successful CAR platform:
ProMab Biotechnologies has a collection of >3,000 antibody products. The high-throughput flow cytometric screening of hybridomas identifies high-binding antibodies for immunotherapeutics.
From the development of an antibody hybridoma, we are able to sequence and isolate the scFv sequence.
The scFv sequence is inserted into a lentiviral vector along with CAR domains of your choosing. High- titer virus particles are prepared and titered by qRT-PCR.
Primary T cells are activated, transduced by the CAR-encoding lentivirus and expanded in culture for two weeks. ProMab has generated >30 types of CAR-T cells and target cells.
The RTCA assay provides highly sensitive real-time measurements of CAR-T cell cytotoxicity against target cells at different E:T ratios. CAR-T cells produce IFN-γ and other cytokines in response to target cells.
Label-free high-throughput Octet ForteBio binding assays. Affinity maturation and humanization of antibodies are available to increase antibody affinity. Bioinformatics and computer modeling generate humanized and affinity-tuned antibodies.
NSG/NOG xenograft survival and imaging studies characterize CAR-T cells in vivo. Toxicology tests measure the toxicity of the CAR-T cells in vivo.
ProMab manufactures QA-validated CAR-T cells and access to GMP facilities.
|
|